What Kind Of Covalent Bonds Does Oxygen Form
If this were the configuration used in covalent bonding carbon would only be able to form two bonds. Chemistry End of Chapter Exercises.

Covalent Bond In Molecules O Level Chemistry Notes
Because they are relatively small carbon atoms can come close enough together to form strong CC double bonds or even C C triple bonds.
What kind of covalent bonds does oxygen form. Carbon also forms strong double and triple. Fluorine is a pale yellow gas that reacts with most substancesThe free element melts at 220 C and boils at 188 CFinely divided metals burn in fluorine with a bright flameNineteen grams of fluorine will react with 10 gram of hydrogen. Electronegativity is a measure of an atoms ability to attract shared electrons to itself.
Unsaturated hydrocarbons are hydrocarbons that have double or triple covalent bonds between adjacent carbon atomsThe term unsaturated means more hydrogen atoms may be added to the hydrocarbon to make it saturated ie. As a result the most electronegative elements are found on the top right of the periodic table while the least electronegative elements are found on the. Any substance that contains only one kind of an atom is known as an elementBecause atoms cannot be created or destroyed in a chemical reaction elements such as phosphorus P 4 or sulfur S 8 cannot be broken down into simpler substances by these reactions.
In reality however the bonds in most substances are neither purely ionic nor purely covalent but lie on a spectrum between these extremes. Classify the six underlined properties in the following paragraph as chemical or physical. Carbon has a valence of 4 which means it bonds with pretty much anything it encounters plus it bonds with more than one thing at a time which makes it the whore of chemistry if you want to look at it that way.
In this case the valence shell would have six electrons- two shy of an octet. Chapter 5 Covalent Bonds and Introduction to Organic Molecules Chemical bonds are generally divided into two fundamentally different types. Water decomposes into a mixture of hydrogen and oxygen when an electric current is passed through the liquid.
Consisting all single bonds. Carbon therefore forms covalent bonds with a large number of other elements including the hydrogen nitrogen oxygen phosphorus and sulfur found in living systems. For many molecules the sharing of electrons allows each atom to attain the equivalent of a full valence.
However the tetrahedral structures of methane and carbon tetrachloride demonstrate that carbon can form four equivalent bonds leading to the desired octet. A covalent bond is a chemical bond that involves the sharing of electron pairs between atomsThese electron pairs are known as shared pairs or bonding pairs and the stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding. On the periodic table electronegativity generally increases as you move from left to right across a period and decreases as you move down a group.
The configuration of an unsaturated carbons include straight chain such as alkenes and alkynes as well as branched chains and aromatic compounds.

Covalent Bonding In An Oxygen Molecule Covalent Bonding Chemistry Worksheets Chemistry Classroom

Covalent Compounds Manoa Hawaii Edu Exploringourfluidearth
Covalent Bond Ck 12 Foundation
How Many Covalent Bonds Can Hydrogen Oxygen Nitrogen And Carbon Form And Why Quora
How Many Covalent Bonds Can Be Formed By Atoms Of Carbon Nitrogen Oxygen And Hydrogen Quora
Learn About Covalent Bonding Chegg Com

Covalent Bonding Biology Definition Role Expii
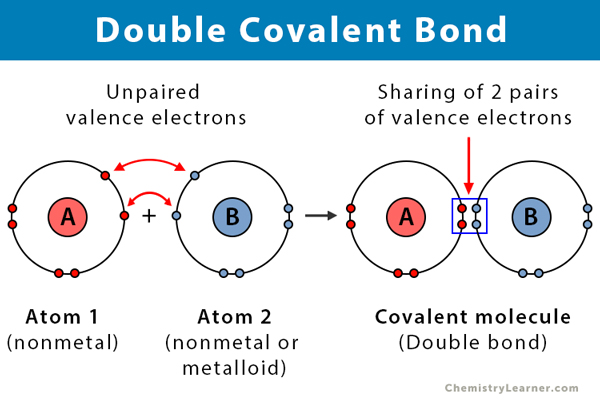
Double Covalent Bond Definition And Examples

Why Is It Possible For An Oxygen Atom To Form A Double Covalent Bond But It Is Not Possible For A Chlorine Atom To Form A Double Covalent Bond Enotes Com

Chemical Bonds Anatomy And Physiology I

Oxygen Molecule O2 Lewis Dot Cross Electronic Diagram Covalent Bonds Ball Stick Space Filling 3d Models Boiling Point Melting Point Doc Brown S Chemistry Revision Notes

Chemical Bonding Chemistry Grade 10 Caps Openstax Cnx

How To Predict Number Of Bonds Each Element Forms Chemsimplified

Reading Covalent Bonds Biology I
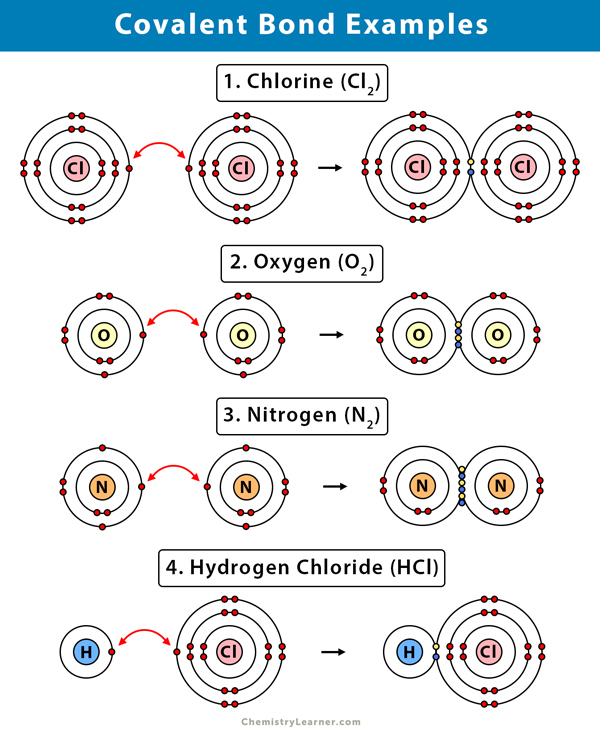
Covalent Bond Definition Types And Examples
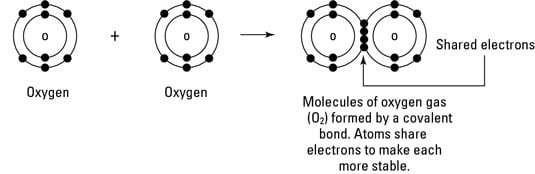
Environmental Science What Is Covalent Bonding Dummies
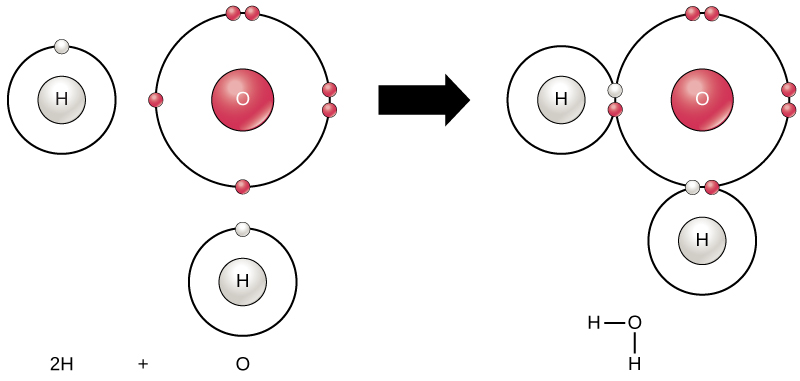
Chemical Reactions And Molecules Biology For Majors I

